
Software Tool Validation in the Medical Device Industry – A White Paper
WHITE PAPER ON REGULATORY REQUIREMENTS AND BEST PRACTICES FOR VALIDATION For More News on Validation:

Agile eQMS to Move Faster & Break Nothing
Our customer Orthogonal describes how to set up an agile eQMS solution on Atlassian Confluence and Jira

In Search of a Compliance Solution that Lets You Sleep at Night
How to find the best regulatory compliance solution for medtech on Atlassian

SoftComply Risk Manager vs Risk Manager Plus on Jira Cloud
Comparison between the risk management apps on Atlassian Jira - the SoftComply Risk Manager and the Risk Manager Plus on Jira Cloud

Software Validation: A Comprehensive Approach to Software Validation in the Medical Device Industry
Automated Validation of Confluence Cloud

On the New FDA Guidance on Software Assurance
On September 13 2022, the FDA issued a new draft guidance on “Computer Software Assurance for…

How to Maintain a Documentation Baseline of Agile Development in Confluence?
most popular use cases for capturing and freezing the always dynamic data in Confluence with the SoftComply Static Snapshots app

Change Management & Medical Device QMS on Confluence
How to keep your QMS up to date when regulations and standards change over time with the help of a Compliance Matrix

Display Document Approvals on Confluence Pages with the SoftComply Change History app
display your document approval history on Confluence pages with the SoftComply Change History app

Introducing the Cloud eQMS Solution for Confluence
Why Confluence? Or perhaps the first question should be – why quality management? Most medical device…
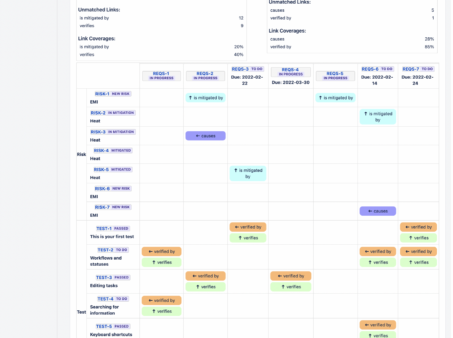
How to Create a Risk Traceability Matrix in Jira
Introduction Risk Traceability is a great way to get a quick overview of the completeness of risk…
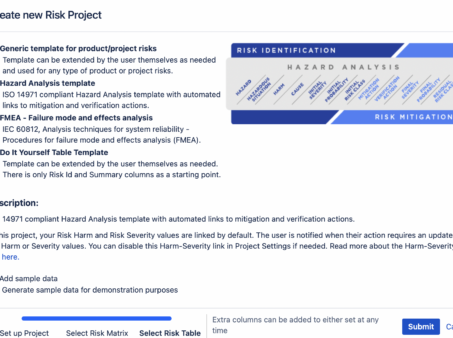
Risk Management in Jira: Streamlining the Process with Powerful Tools
Risk Management in Jira In today’s fast-paced and ever-evolving business landscape, effective risk management plays a…