
How to Make your Confluence Pages Static & Compare them with the SoftComply Static Snapshots app
Introduction With the SoftComply Static Snapshots app you can capture and freeze the dynamic content of…

Six Must-Have Atlassian Cloud Apps for the Regulated Industries
Introduction With Atlassian’s Server end of life approaching in a few years, most companies need to…

How to Manage Requirements in Confluence and Jira
Introduction to Requirements Management in Jira and Confluence Requirements management in the regulated domains such as…
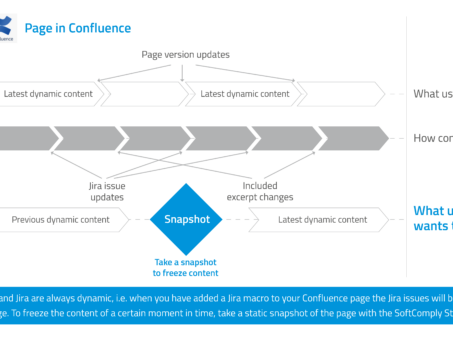
How to keep your data in Confluence static?
If you have worked with Jira and Confluence, am sure you have enjoyed the integration between…
Risk Management Guide for a Digital Health or a Medical Device Company on Jira Cloud
Why Risk Management In the fast-paced world of medical device development, safety and quality are of…

How to Build a Risk Analysis in Jira
How to build a risk analysis in Jira with the SoftComply Risk Manager app

Compliance of myBioma
The Regulatory Compliance Journey of myBioma Biome Diagnostics GmbH is an Austrian medtech start-up utilizing the…

3 MUST-HAVE AREAS TO CONSIDER WHEN SETTING YOUR ORGANIZATION UP ON ATLASSIAN STACK
3 things regulated industries must consider when setting up their organisations on Atlassian Jira and Confluence
SoftComply
How to apply SoftComply Risk Manager features to any Jira project
Wouldn’t it be cool to keep everything in Jira – your backlog, your test cases and…
SoftComply
How to Add Page Numbers to the PDF Exports of Quality Records in Confluence
Paper-based Quality Management Systems are now (almost) a thing of the past (it’s sooo 20th century…).…

Medical Device Compliance & Atlassian Cloud
On the 16th of October 2020, Atlassian announced radical changes to their offering. From February 2nd…

How We Managed the Compliance of Personal Protective Equipment
The Regulatory Compliance Journey of XRGO Nathaniel Victor, CEO of XRGO: “We got involved in PPE…