Six Must-Have Atlassian Cloud Apps for the Regulated Industries

Introduction With Atlassian’s Server end of life approaching in a few years, most companies need to decide whether to migrate to Cloud or to Data Center. Data Center may seem like a natural path to take for the larger (over 300 users) companies in the regulated domains. Nevertheless, there are some regulated industries like the […]
How to Manage Requirements in Confluence and Jira

Introduction to Requirements Management in Jira and Confluence Requirements management in the regulated domains such as medtech, automotive or aviation has certain differences compared to the agile development. Jira Software with its easy to use backlog view and work boards suits best for managing requirements in the agile world. For the regulated domains it might […]
How to keep your data in Confluence static?
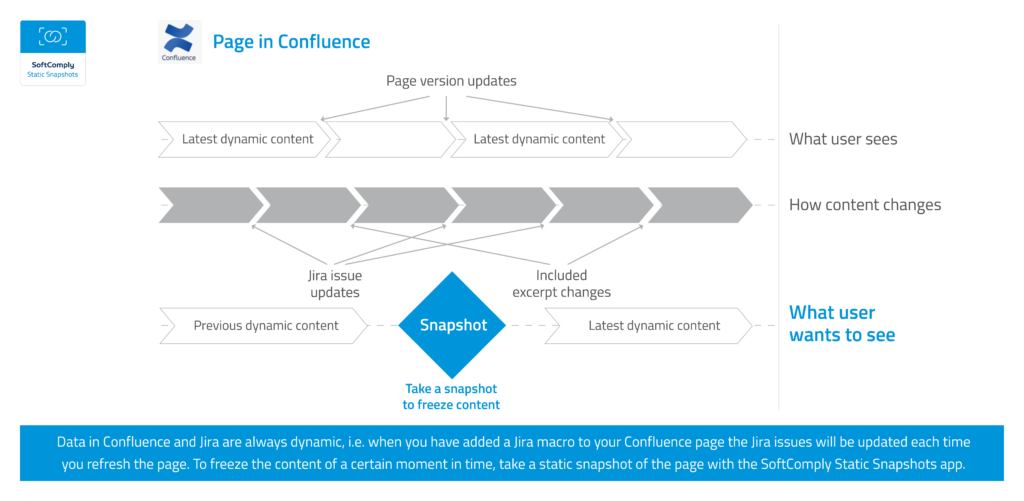
If you have worked with Jira and Confluence, am sure you have enjoyed the integration between the two. The way you can easily pull data from Jira to Confluence by creating macros on your Confluence page with all the data from Jira that you want to display is absolutely wonderful. Now, every time you refresh […]
Relationship between FDA & ISO 13485

The relationship between the FDA and ISO 13485 goes back a long time. The ISO standard has been in the list of General Consensus Standards since the dawn of times, but there has never been a full alignment between 21 CFR 820 (QSR) and ISO 13485. It’s like these couples that have been together forever, […]
Compliance of myBioma

The Regulatory Compliance Journey of myBioma Biome Diagnostics GmbH is an Austrian medtech start-up utilizing the genetic information of the microbiome and AI to develop medical diagnostic software for doctors. Biome Dx products utilize technologies such as next-generation sequencing, pipeline architectures and microservices. With their lifestyle product myBioma every European has the possibility to learn […]
How to Add Page Numbers to the PDF Exports of Quality Records in Confluence
Paper-based Quality Management Systems are now (almost) a thing of the past (it’s sooo 20th century…). But in some cases, printing records and documents from the eDMS is necessary, or at least exporting them to PDF. The requirements for paper documents are the same as digital documents. But, unlike electronic formats, physical misplacement or loss […]
Medical Device Compliance & Atlassian Cloud

On the 16th of October 2020, Atlassian announced radical changes to their offering. From February 2nd 2021, Atlassian will not sell any new Server licenses for any of their products. A year later, in February 2022, it will not be possible to upgrade or downgrade your Atlassian Server product tier. From February 2023, it will […]
How We Managed the Compliance of Personal Protective Equipment

The Regulatory Compliance Journey of XRGO Nathaniel Victor, CEO of XRGO: “We got involved in PPE manufacturing as a goodwill donation/gesture of time, energy, products and financial support to the hospitals and local municipalities. Early on, our suppliers in China informed us that COVID-19 was serious and they advised that we protect our staff and […]
How to Use Confluence Pages as Templates
Summary SoftComply has released a new Confluence Server macro within the SoftComply eQMS app that allows the user to use any Confluence page as a template, copying it automatically into the existing space at a click of a button. Features include the addition of prefixes, suffixes, timestamp and more. This bypasses the use of Confluence […]
How Cibiltech became FDA 21 CFR 11 Compliant on Atlassian Confluence

Customer Story on the MediCompli Solution at Cibiltech NOTE: Cibiltech has now successfully migrated from Atlassian Server to Atlassian Cloud and have implemented the Cloud eQMS Solution as their Quality Management Solution. Cibiltech Cibiltech is a French medical device company developing an AI based personalized medicine solution for better patient care. Their most recent solution […]
