Risk Manager
SoftComply Risk Manager is a Jira app to manage, trace and report your software product and project risks. It is is fully customizable and integrated with Atlassian Jira.





Build your Risk Model
Use the ready-made templates of Risk Matrix or RPN based Risk Models or build your own Risk Model from scratch.
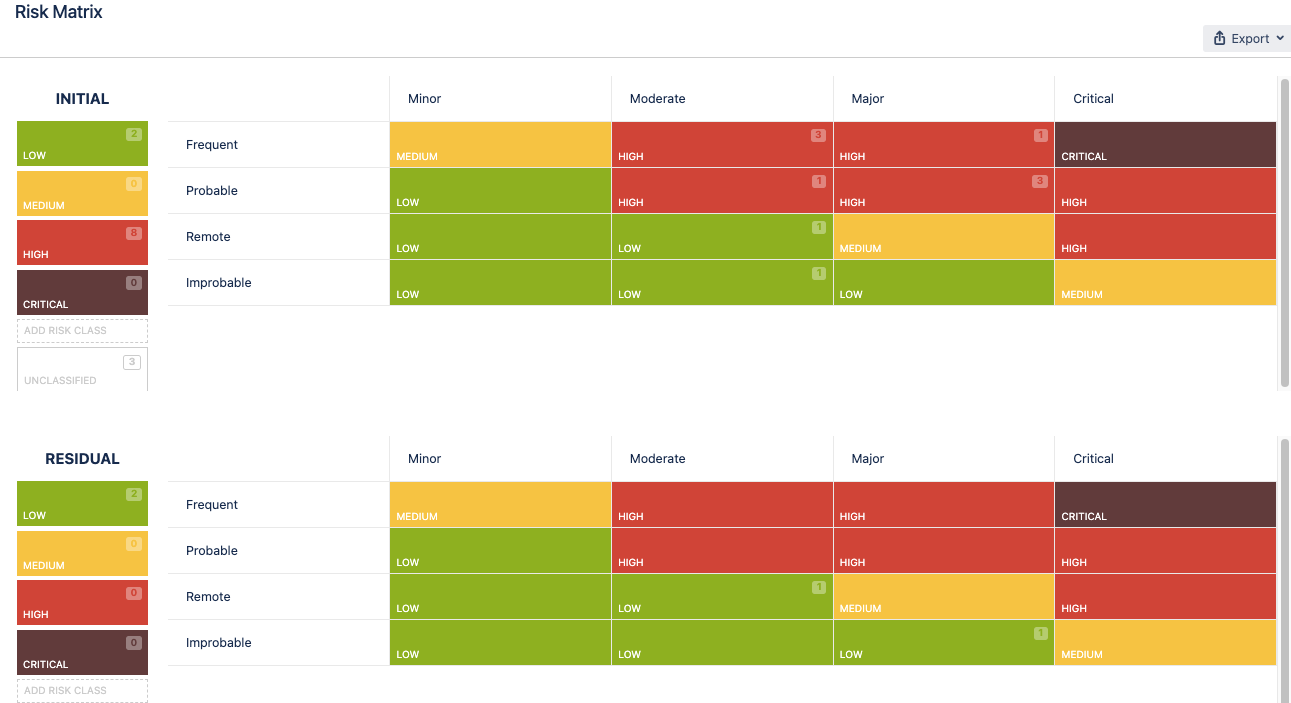
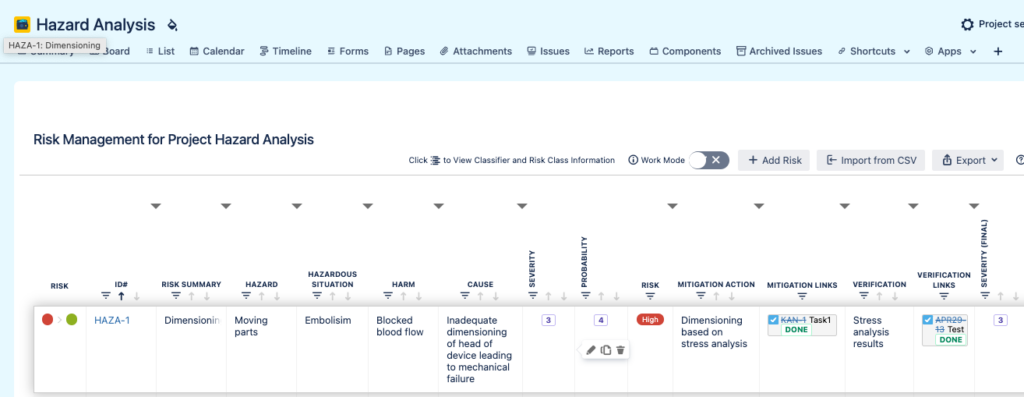
Manage Product or Project Risks in a Table Format
Use an Excel-like table inside Jira to manage your project or product risks.
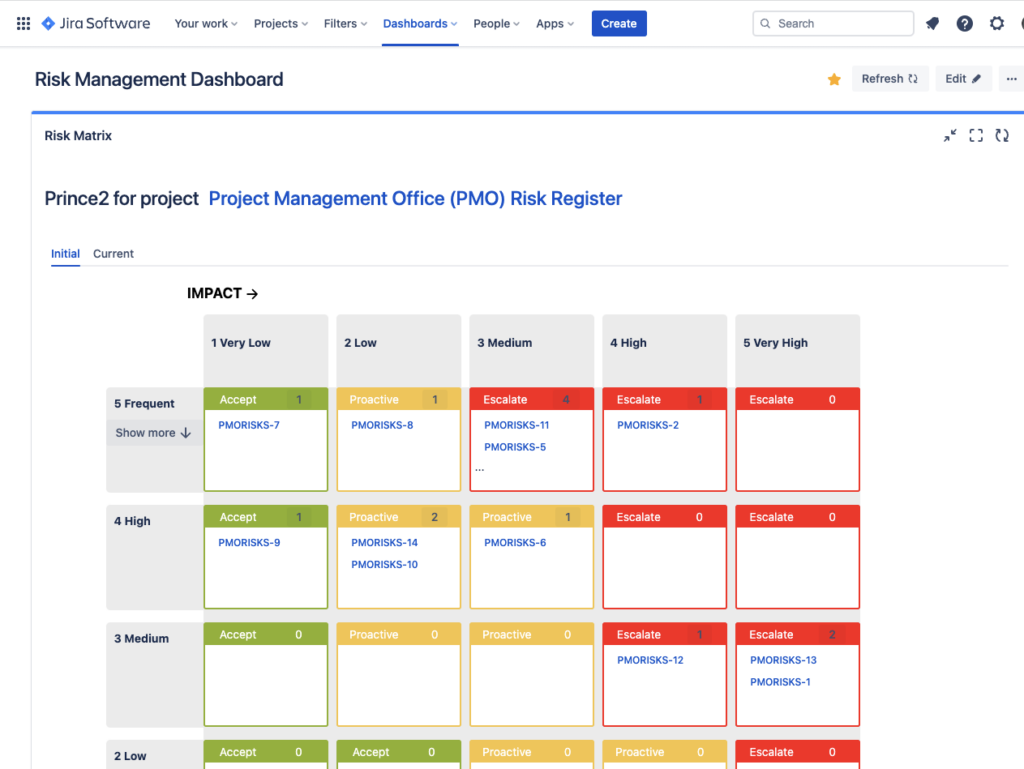
Report Risks in Dashboards
You can easily report your project or products risks in Jira Dashboard.
Why use SoftComply plugins

No More Disconnected Data
SoftComply apps are native to Jira and Confluence, so all your data and processes are connected.

Dedicated Interface
Our apps are built on Jira and Confluence – but they are unique tools with dedicated interfaces.

Quick and Easy Setup
Get started fast with built-in templates and embedded guidance.

Atlassian Gold Partner
SoftComply is a trusted Atlassian Gold Partner. Count on us for reliability and security – we’ve been vetted and validated by Atlassian's rigorous approval standards.

Top-Rated by Users
Our apps are top-rated on the Atlassian Marketplace – trusted by SoftComply users. Risk Manager Plus has a near-perfect rating of 3.9 out of 4 stars.

10x Cheaper
SoftComply apps let you leverage your existing time and financial investment in Atlassian. You get the power of a standalone solution in an affordable Jira-or Confluence-based app.
See Risk Manager in action
What our clients say
The Risk Manager is an excellent app from SoftComply
The Risk Manager supports Risk Management for both Jira Service management and Jira Software! I would definitely recommend it to other companies.
Pricing
Resources
Why can’t I create a new Risk Project?
There could be several reasons for this. Please have a look at your SoftComply Risk Manager configuration and answer the following questions here.
Why did my Risk Severity values change?
This is a feature of the SoftComply Risk Manager app that is related to the requirement of ISO 14971 for medical device risk management. To learn more about it and how to disable this option, please continue reading here.
Why do I see empty Risk Rows in the Risk Table?
For more information about why you may be seeing empty risk rows in the risk management table and how to avoid it, please continue reading here.
Which permissions do I need for the SoftComply Risk Manager?
Please read the Security and Permissions related questions and answers at the Risk Manager Security post.
Latest videos and news
SoftComply
@softcomply · 253 subscribers · 145 videos

SoftComply
@softcomply · 253 subscribers · 145 videos

👉 Try SoftComply Risk Manager Plus free for a month: https://marketplace.atlassian.com/apps/1219692/softcomply-risk-manager-plus-top-risk-management-in-jira?tab=overview&hosting=cloud 👉 Book a live demo: https://calendly.com/softcomply/softcomply-intro?_gl=1%2A1isw2n2%2A_ga%2AMTE2NDUyODgwOS4xNzE5MTI2MzY4%2A_ga_CSPTE6LB5Z%2AMTcyMDUzODExNi42LjAuMTcyMDUzODExNi42MC4wLjIwMDkwODcxMw..&month=2024-07
Subscribe to the SoftComply Blog
Stay connected with SoftComply and receive new blog posts in your inbox.
Ready to get started?
Contact us to book a demo and learn how SoftComply can cover all your needs

