Risk Management in Jira: Streamlining the Process with Powerful Tools
Risk Management in Jira In today’s fast-paced and ever-evolving business landscape, effective risk management plays a…
How to Report Risks in Confluence
Introduction Managing your product risks in Jira is great – you can use the flexibility of Jira…
Risk Management Guide for a Digital Health or a Medical Device Company on Jira Cloud
Why Risk Management In the fast-paced world of medical device development, safety and quality are of…

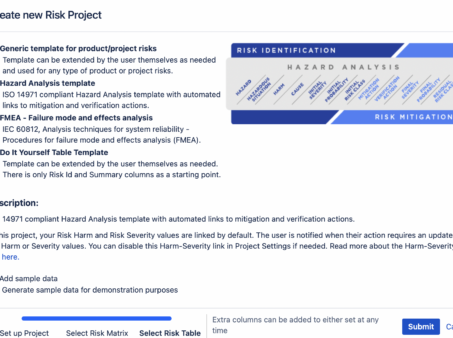
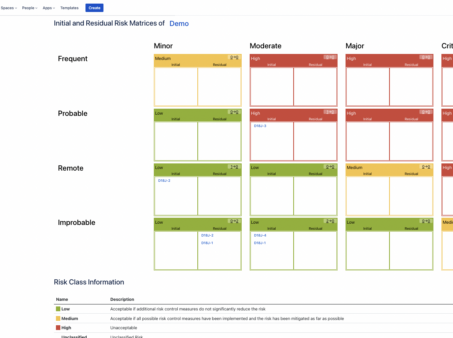
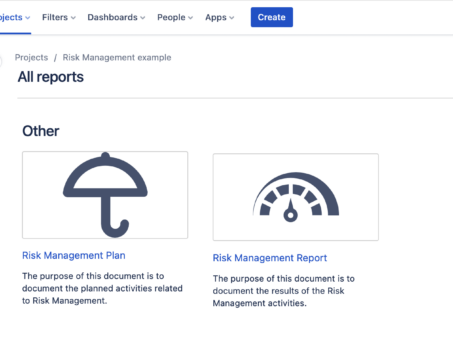
How to Build a Risk Analysis in Jira
How to build a risk analysis in Jira with the SoftComply Risk Manager app

Compliance of myBioma
The Regulatory Compliance Journey of myBioma Biome Diagnostics GmbH is an Austrian medtech start-up utilizing the…
SoftComply
How to apply SoftComply Risk Manager features to any Jira project
Wouldn’t it be cool to keep everything in Jira – your backlog, your test cases and…
SoftComply
Why Decompose the Probability of Harm in two – P1 and P2
P1 and P2 No we are not talking about the early Playstation versions… ISO 14971 (2019…
SoftComply
What are the best Risk Management Apps on Jira & how they compare to each other?
In the following post we compare the features of 4 top risk management apps on Jira…
SoftComply
Risk Management for JIRA. Why?
Compliant Risk Management is a mandatory regulatory requirement for companies in medical device and other safety-critical…

What is FMEA and how is it different from Hazard Analysis?
From ISO 14971: “FMEA is a technique by which the consequences of an individual fault mode…
SoftComply
How I came to hate Excel & decided to develop an automated Risk Management tool for JIRA
Part II By Matteo Gubellini, VP of Regulatory Affairs at SoftComply* So you are looking for…
SoftComply
How I came to hate Excel & decided to develop an automated Risk Management tool for JIRA
Part I By Matteo Gubellini, VP of Regulatory Affairs of SoftComply* “Ok, let’s follow a few…